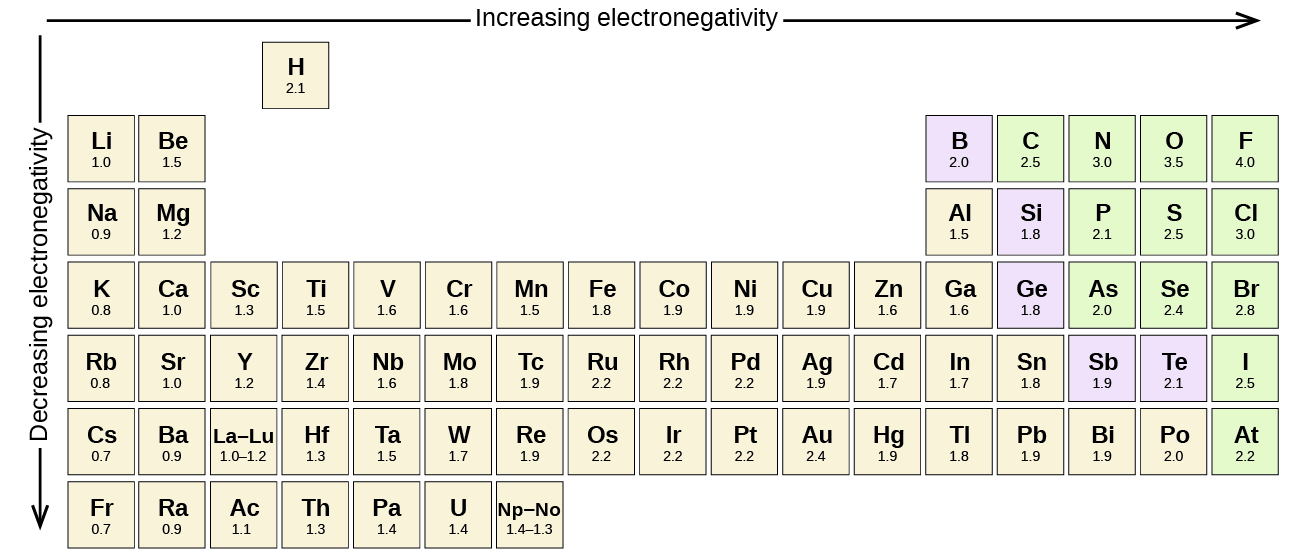 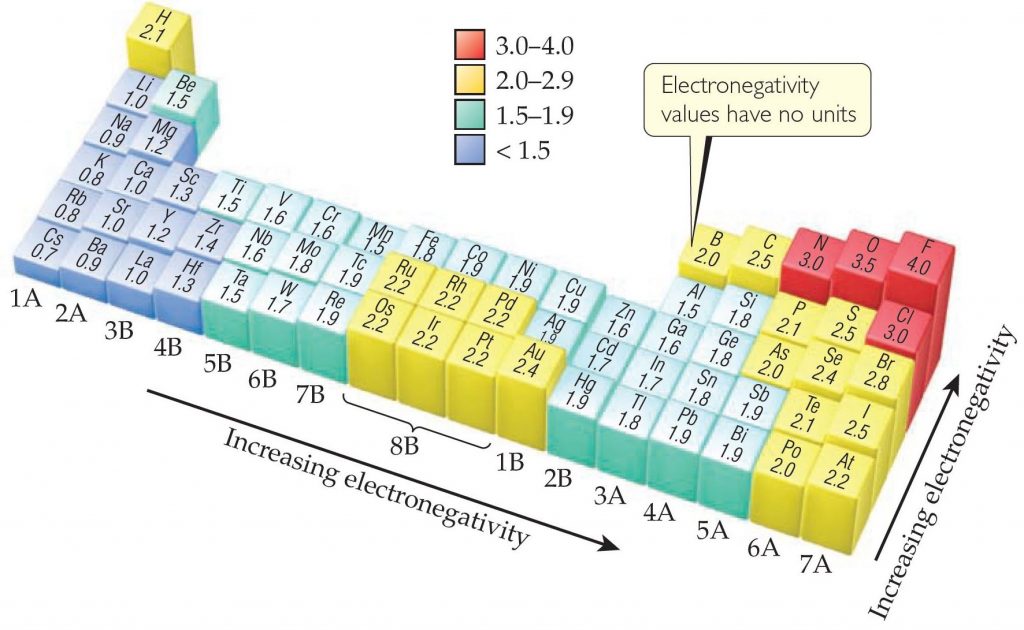 
Carbon is more electronegative than you think. 
This is a good example of something that is worth the time to brute-force memorize. Just knowing these 12 numbers will take you a long way. From the concepts that spring from the facts in this table come many, many potential exam questions. Here is a table of the electronegativities of the 12 elements you will likely encounter the most. In organic chemistry, we tend to focus on elements that form covalent bonds to carbon and hydrogen. If you are just starting out in organic chemistry, let me say it now: electronegativity rears its head in organic chemistry in more ways than you can currently imagine.The direct and indirect effects of electronegativity dictate important properties such as solubility, acidity, hydrogen bonding,melting and boiling points, chemical reactivity and many more. The resulting dipoles play a tremendous role in organic chemistry. Like in many human relationships, when elements get together to form bonds, a sort of hierarchy is established: in this case, the more electronegative element gets an unequal share of the electrons. At the bottom left corner of the periodic table lies Cesium, which lacks a catchy nickname with an electronegativity of 0.79. Fluorine, the “ Tiger of Chemistry“, is head of the class at 4.0. What makes it great is its simplicity: like GPA, it is a single number that gives an immediate mental impression of an element’s personality. In a nutshell, it is a measure of how “hungry” an element is for electrons, which is a function of the number of electrons in the valence shell and the effective nuclear charge felt by them. Excellence in solving chemistry queries in excels and word format.Electronegativity is one of the most useful concepts in chemistry.Quality solution with 100% plagiarism free answers.Higher degree holder and experienced experts network.These are avail for subscribed users and they can get advantages anytime. We prepare quality content and notes for Variation of electronegativity in the periodic table topic under chemistry theory and study material. We provide step by step Variation of electronegativity in the periodic table question's answers with 100% plagiarism free content. Live tutors are available for 24x7 hours helping students in their Variation of electronegativity in the periodic table related problems. Pauling and Mulliken values of electronegativities are related as below (Pauling) = 0.34 (Mulliken) - 0.2Įmail based Chemistry assignment help - homework help at ExpertsmindĪre you searching chemistry expert for help with Variation of electronegativity in the periodic table questions? Variation of electronegativity in the periodic table topic is not easier to learn without external help? We at finest service of Chemistry assignment help and chemistry homework help. Where and are electronegativities of the atoms A and B respectively, the factor 0.208 comes from the conversion of kcal to electron volt (1 eV = 23.0 kcal/mole), He described electronegativity difference between the two atoms and then by assigning arbitrary values to few elements ( for example 4.00 to fluorine, 2.5 to carbon and 2.1 to hydrogen), he then calculated electronegativity of the other elements. This is based on the excess of bond energies. (iii) Pauling scale : Pauling scale of electronegativity is the most widely used. Thus where Z is the effective nuclear charge and r is the covalent radius of the atom in Å. (ii) Allred-Rochow scale : Allred and Rochow defined electronegativity as the electrostatic force exerted by the nucleus on the valence electrons. (i) Mulliken's scale : Mulliken regarded electronegativity as the average value of ionization potential and electron affinity of an atom. (3) Electronegativity can be expressed on the following three scales If an element exhibits various oxidation state, the atom in higher oxidation state will be more negative because of greater attraction for the electron, for example Sn II (1.30) and Sn IV (1.90). This is because of increase in atomic size. (ii) In a group, electronegativity decreases from top to bottom. The inert gases posses zero electronegativity. Therefore the alkali metals have the lowest value, while the halogens have the highest. This is because of decrease in size and increase in nuclear charge. (i) In the period, electronegativity increases from left to right. The variation of electronegativity in the periodic table
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
